Here’s a question: why do you choose to eat certain foods at certain times, and not other things at other times? Why will a mouse work so hard for a sugar reward, and why does it keep coming back? Moreover, why does it ever stop eating? New research in flies is beginning to uncover how they detect food sources in the environment and evaluate their nutritional values, and how they decide whether or not to dive in and chow down.
Animals employ a diverse arsenal of tools to tell whether a food source is worth pursuing. A fly’s sense of smell helps it to locate a piece of rotting fruit and fly towards it. Once it gets there, the fly uses its sense of taste to what the food contains: sugars, toxins, salts and so on. But these senses are not completely infallible: artificial sweeteners, for example, taste great but have very little nutritional value. It is only in the last few years that we have begun to see that even after eating a food, animals are capable of assessing its nutritional value, assigning this value to a particular food source, and using this value in future decisions about what to eat. What’s more, we are getting closer to understanding what an internal nutrient sensor really is, and how it is integrated with other senses to guide feeding decisions.
Last year, three groups independently demonstrated that flies can detect the nutritional content of food independently of its taste. Dus et al (2011) allowed flies to make a choice between two different food sources – one containing red dye, and the other blue – and assessed their food choices based on the colour of the flies’ abdomens after 2 hours.
Given a choice between a nutritious sugar (sucrose) and agar containing no food source, flies will usually choose sucrose. The researchers then made use of mutant flies that lack the ability to detect external sugars, due to an absence of the taste receptor molecules Gr5a and Gr64a, or due to an absence of all taste receptor organs. After 5 hours of starvation, these flies eat much less sugar, and many more of them eat nothing at all, since they cannot taste the sucrose. However, after 22 hours of starvation, these flies eat just as much sucrose as normal flies. This works for different sugars, such as fructose and glucose, but not for non-nutritious D-glucose or sucralose. This suggests that in a starved state, the flies rely much more on their internal assessment of a food’s nutritional benefits, rather than an external sense of taste, to guide their choices.
Fujita & Tanimura (2011) and Burke & Waddell (2011) both both demonstrate that flies can learn an attraction to a food source based purely on its nutritional value, without the attraction of sweet taste. If an odour is presented together with a sweet-tasting food, flies will rapidly learn to approach this odour when it reappears. Fujita & Tanimura showed that flies can learn such an association even when the sugar is tasteless (sorbitol), as long as it is nutritious. Burke & Waddell used a similar odour-pairing test, and showed that while short-term memory is based mainly on sweet taste – less nutritious sweeteners such as arabinose were as effective as fructose as rewards – long-term odour memories are formed much more effectively by sugars that provide a nutritional benefit. While long-term memories formed by non-nutritious but sweet arabinose are very weak, supplementing this sugar with nutritious but tasteless sorbitol allows the formation of strong memories, showing that flies can assess the nutritional content of a food after eating it and use this to guide future decisions.
To understand how a fly integrates this nutritional information into its picture of a food source, Stafford et al (2012) made use of a fly café. The CAFE (CApillary FEeding assay) is not like your regular greasy spoon: the flies are provided with tubes extending from their ceiling, each containing liquid fly food. As the levels of the liquids change, one can track the amount of food eaten by the six-legged customers. They showed that flies’ initial food choices can be explained by their taste alone, but that over time their consumption shifts to reflect the nutritional content of the food sources. For example, given the choice between a sweet, non-nutritious sugar and a less sweet but highly nutritious sugar, the flies initially choose the former, but over time they increasingly choose the latter. Indeed, this shift to nutritious sugars occurs much more rapidly in hungry flies, beginning within 20 minutes. So it seems that the role of the internal nutrient sensor is to refine the initial evaluations of food sources by the taste system. But still, the identity of this internal nutrient sensor remains elusive..
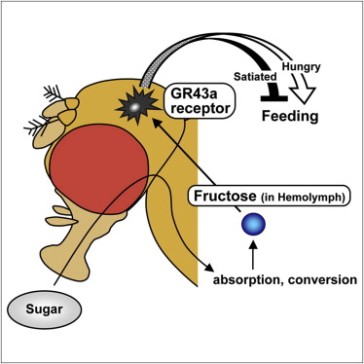
… Or at least it did, until last week. In the most recent issue of Cell, Miyamoto et al identify a molecule essential to the functioning of an internal sugar sensor in flies. First, they show that the taste receptor Gr43a is a specific receptor for fructose, a sugar found, surprisingly, in fruits. As well as in taste organs, Gr43a is also found in flies’ brains. Flies lacking Gr43a in the brains cannot base their food choices on the nutritional sugar content of foods, but rely on their perceived sweetness alone. Thus, flies detect changes in the level of fructose in the haemolymph (fly blood) following a meal as a measure of the food’s nutritional content. But the main sugars in the haemolymph are trehalose and glucose – so why use fructose? Well, the levels of trehalose and glucose have to be very tightly regulated, to keep their levels stable so that all organs receive just the right amounts of each. But fructose, as a minor constituent of the haemolymph, is free to vary much more widely, so it serves as a much more reliable indicator of the amount of sugar entering the body from a meal.
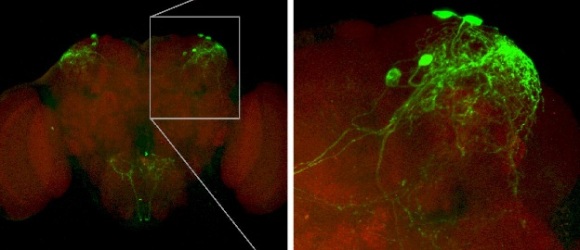 Gr43a is found in only 6-8 neurons of the brain (out of around 100 000 neurons in all). Miyamoto et al show that the effect of activating these neurons depends on the state of the fly. They use genetically implanted channels that render just these few neurons sensitive to temperature, so that by raising the temperature they activate these neurons at specific times. A similar odour-pairing test is used as above – one odour is paired with activation of these neurons, while the other odour remains neutral, and the fly is then given a choice between the two.
Gr43a is found in only 6-8 neurons of the brain (out of around 100 000 neurons in all). Miyamoto et al show that the effect of activating these neurons depends on the state of the fly. They use genetically implanted channels that render just these few neurons sensitive to temperature, so that by raising the temperature they activate these neurons at specific times. A similar odour-pairing test is used as above – one odour is paired with activation of these neurons, while the other odour remains neutral, and the fly is then given a choice between the two.
These experiments show that in full flies, activation of these neurons is perceived as negative, and they will avoid activating them. In contrast, hungry flies prefer to activate the neurons. In practice, this would mean that fed flies would avoid eating nutritious foods, while hungry flies would eat in order to raise their fructose levels and activate these neurons. So not only do these neurons act as internal nutrient sensors of sugars in the haemolymph using Gr43a, but activating them can be either rewarding or repulsive, depending on the feeding state of the fly, and thus they can guide useful feeding decisions to keep the fly well-fed but prevent overeating. Now if only we humans could make good decisions like those. We have a lot to learn from the humble fruit fly.
Dus M, Min S, Keene AC, Lee GY & Suh GSB (2011). Taste-independent detection of the caloric content of sugar in Drosophila. PNAS 108, 11644–11649.
Fujita M & Tanimura T (2011). Drosophila Evaluates and Learns the Nutritional Value of Sugars. Current Biology 21, 751–755.
Burke CJ & Waddell S (2011). Remembering nutrient quality of sugar in Drosophila. Curr Biol 21, 746–750.
Stafford JW, Lynd KM, Jung AY & Gordon MD (2012). Integration of taste and calorie sensing in Drosophila. J Neurosci 32, 14767–14774.
Miyamoto T, Slone J, Song X & Amrein H (2012). A Fructose Receptor Functions as a Nutrient Sensor in the Drosophila Brain. Cell 151, 1113–1125.